Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tang, J.*; Seo, O.*; Rivera Rocabado, D. S.*; Koitaya, Takanori*; Yamamoto, Susumu*; Namba, Yusuke*; Song, C.*; Kim, J.*; Yoshigoe, Akitaka; Koyama, Michihisa*; et al.
Applied Surface Science, 587, p.152797_1 - 152797_8, 2022/06
Times Cited Count:7 Percentile:75.56(Chemistry, Physical)The hydrogen absorption and diffusion mechanisms on cube-shaped Pd nanoparticles (NPs) which are important hydrogen-storage materials were studied using X-ray photoelectron spectroscopy and DFT calculations. In the surface region, hydrogen absorption showed almost similar behavior regardless of the NPs size. It was found that the octahedral sites are more favorable than the tetrahedral sites for hydrogen occupation. We also clarified that the hydrogen atoms absorbing on the smaller-sized Pd NPs diffuse to the subsurface more actively because of the weakened Pd-H bond by the surface disordering, which plays an important role in hydrogen adsorption at a low H pressure.
pressure.
Fujita, Yoshitaka; Niizeki, Tomotake*; Fukumitsu, Nobuyoshi*; Ariga, Katsuhiko*; Yamauchi, Yusuke*; Malgras, V.*; Kaneti, Y. V.*; Liu, C.-H.*; Hatano, Kentaro*; Suematsu, Hisayuki*; et al.
Bulletin of the Chemical Society of Japan, 95(1), p.129 - 137, 2022/01
Times Cited Count:8 Percentile:74.08(Chemistry, Multidisciplinary)In this work, the mechanisms responsible for the adsorption of molybdate ions on alumina are investigated using in-depth surface analyses carried out on alumina specimens immersed in solutions containing different molybdate ions at different pH values. The obtained results reveal that when alumina is immersed in an acidic solution containing molybdate ions, the hydroxyl groups present on the surface are removed to generate positively charged sites, and molybdate ions (MoO
 or AlMo
or AlMo O
O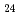 H
H
 ) are adsorbed by electrostatic interaction. Alumina dissolves slightly in an acidic solution to form AlMo
) are adsorbed by electrostatic interaction. Alumina dissolves slightly in an acidic solution to form AlMo O
O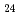 H
H
 , which is more easily desorbed than MoO
, which is more easily desorbed than MoO
 . Furthermore, the enhancement in the Mo adsorption or desorption property may be achieved by enriching the surface of the alumina adsorbent with many -OH groups and optimizing Mo solution to adsorb molybdate ions on alumina as MoO
. Furthermore, the enhancement in the Mo adsorption or desorption property may be achieved by enriching the surface of the alumina adsorbent with many -OH groups and optimizing Mo solution to adsorb molybdate ions on alumina as MoO
 ions. These findings will assist researchers in engineering more efficient and stable alumina-based adsorbents for molybdenum adsorption used in medical radioisotope (
ions. These findings will assist researchers in engineering more efficient and stable alumina-based adsorbents for molybdenum adsorption used in medical radioisotope ( Mo/
Mo/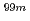 Tc) generators.
Tc) generators.
 -trimethylglycine
-trimethylglycineSuzuki, Tomoya*; Narita, Hirokazu*; Ogata, Takeshi*; Suzuki, Hideya; Matsumura, Tatsuro; Kobayashi, Toru; Shiwaku, Hideaki; Yaita, Tsuyoshi
Solvent Extraction Research and Development, Japan, 26(1), p.11 - 19, 2019/06
Times Cited Count:3 Percentile:13.73(Chemistry, Multidisciplinary)The ability of AMP03, a styrene-divinylbenzene copolymer functionalized with  -trimethylglycine moieties, to adsorb Pd(II) from HNO
-trimethylglycine moieties, to adsorb Pd(II) from HNO solutions was investigated to elucidate the affinity of
solutions was investigated to elucidate the affinity of  -trimethylglycine for Pd(II). In the present study, we investigated the mechanism of Pd(II) adsorption by AMP03 by means of adsorption experiments, Fourier Transform Infrared (FT-IR) spectroscopy, and Extended X-ray Absorption Fine Structure (EXAFS) spectroscopy.
-trimethylglycine for Pd(II). In the present study, we investigated the mechanism of Pd(II) adsorption by AMP03 by means of adsorption experiments, Fourier Transform Infrared (FT-IR) spectroscopy, and Extended X-ray Absorption Fine Structure (EXAFS) spectroscopy.
Tanaka, Tadao; Mukai, Masayuki; Nakayama, Shinichi
Proceedings of International Conference on Nuclear Energy System for Future Generation and Global Sustainability (GLOBAL 2005) (CD-ROM), 6 Pages, 2005/10
no abstracts in English
Yamaguchi, Tetsuji
Denki Hyoron, 89(8), p.49 - 54, 2004/08
no abstracts in English
Inagaki, Yaohiro*; Mitsui, Seiichiro*; Makino, Hitoshi*; Ishiguro, Katsuhiko*; Kamei, Gento*; Kawamura, Kazuhiro*; Maeda, Toshikatsu; Ueno, Kenichi*; Bamba, Tsunetaka*; Yui, Mikazu*
Genshiryoku Bakkuendo Kenkyu, 10(1-2), p.69 - 84, 2004/03
no abstracts in English
Fumizawa, Motoo; Kameda, Atsuyuki*; Nakagawa, Takashi*; Wu, W.*; Yoshikawa, Hidekazu*
Nuclear Technology, 141(1), p.78 - 87, 2003/01
Times Cited Count:5 Percentile:36.77(Nuclear Science & Technology)no abstracts in English
Uchida, Masahiro; Yoshino, Naoto
JNC TN8410 2001-016, 36 Pages, 2001/05
This technical report summarizes sampling of the natural rock including conductive fracture. Hydraulic test was conducted at the target fracture prior to excavation. Objective of the sample was to reproduce same transmissivity at LABROCK by adjusting normal stress. This report was originally compiled by PNC in october, 1993.
Uchida, Masahiro; Yoshino, Naoto
JNC TN8410 2001-015, 35 Pages, 2001/05
This technical report summarizes excavation and preparation of the natural rock block sample used in LABROCK. This report was originally compiled by PNC in March, 1993.
Tanaka, Tadao; Ogawa, Hiromichi; Muraoka, Susumu
Materials Research Society Symposium Proceedings, Vol.663, p.1169 - 1177, 2001/00
no abstracts in English
; Yoshida, Eiichi; Aoto, Kazumi
JNC TN9400 2000-042, 112 Pages, 2000/03
A sodium-water reaction drove from the single tube break in steam generator of FBR might overheat labor tubes rapidly under internal pressure loadings. lf the temperature of tube wall becomes too high, it has to be evaluated that the stress of tube does not exceed the material strength limit to prevent the propagation of tube rupture. This study clarified the tensile and creep properties of Mod.9Cr-1Mo steel at ultra-high temperature which will be used in evaluation of the tube burst by sodium-water reaction. The strain rates for tensile test are from 10%/min to 10%/sec, and creep-rupture time is maximum 277sec. The range of test temperature is 700 C to 1300
C to 1300 C. The main results obtained were as follows; (1)The evaluation data on the relationship between tensile strength and strain rate and creep-rupture strength in shorter time on Mod.9Cr-1Mo steel were acquired. (2)Short-term mechanical properties of Mod.9Cr-1Mo steel were evaluated based on the results of tensile and creep-rupture tests up to 1300
C. The main results obtained were as follows; (1)The evaluation data on the relationship between tensile strength and strain rate and creep-rupture strength in shorter time on Mod.9Cr-1Mo steel were acquired. (2)Short-term mechanical properties of Mod.9Cr-1Mo steel were evaluated based on the results of tensile and creep-rupture tests up to 1300 C. As a result of the evaluation, recommended equation of creep-rupture strength in the short-term was proposed. (3)Tensile and creep-rupture strength of Mod.9Cr-1Mo steel tube showed the value which was higher than the 2 1/4Cr-1Mo steel, and it was proven to have the superior properties.
C. As a result of the evaluation, recommended equation of creep-rupture strength in the short-term was proposed. (3)Tensile and creep-rupture strength of Mod.9Cr-1Mo steel tube showed the value which was higher than the 2 1/4Cr-1Mo steel, and it was proven to have the superior properties.
Tanaka, Tadao; Ogawa, Hiromichi
Proceedings of the International Workshop on Distribution and Speciation of Radionuclides in the Environment, p.278 - 284, 2000/00
no abstracts in English
Sugino, Hiroyuki; *
JNC TN8400 99-040, 75 Pages, 1999/11
The buffer material that will be buried as a component of the engineered barriers system swells when saturation by groundwater. As a result of this swelling, buffer material may penetrate into the peripheral rock zone surrounding the buffer through open fractures. If sustained for extremely in long-period of time, The buffer material extrusion could lead to reduction of buffer density, which may in turn degrade the assumed performance assessment properties (e.g., permeability, diffusion coefficient) JNC has been conducted the study of bentonite extrusion into fractures of rock mass as a part of high level waste research. In 1997, JNC has reported the test results concerning buffer material extrusion and buffer material erosion. These tests have been done using test facilities in Geological Isolation Basic Research Facility. After 1997, JNC also conducted analytical study of buffer material extrusion. This report describes the analysis results of this study which are reflected to the H12 report. In this analysis, The diffusion coefficient was derived as a function of the swelling pressure and the viscosity resistance of the buffer materials. Thus, the reduction in density of buffer materials after emplacement in saturated rock was assessed. The assessment was made assuming parallel-plate radial fractures initially filled by water only. Because fractures in natural rock masses inevitably have mineral inclusions inside of them and fractures orientation leads to fractures intersecting other fractures, this analysis gives significantly conservative conditions with respect to long-term extrusion of buffer and possible decrease in buffer density.
 S and D
S and D S
SSato, K.*; *; *; *; *; Kurosaki, Yuzuru*; Takayanagi, Toshiyuki
Chemical Physics, 242(1), p.1 - 10, 1999/00
Times Cited Count:7 Percentile:22.54(Chemistry, Physical)no abstracts in English
 Np,
Np, Pu and
Pu and  Am in sedimentary materials
Am in sedimentary materialsTanaka, Tadao; Muraoka, Susumu
Journal of Radioanalytical and Nuclear Chemistry, 240(1), p.177 - 182, 1999/00
Times Cited Count:12 Percentile:66.07(Chemistry, Analytical)no abstracts in English
 Np and
Np and  Am in sedimentary geological formation
Am in sedimentary geological formationTanaka, Tadao; Takebe, Shinichi; Ogawa, Hiromichi; Muraoka, Susumu
JAERI-Research 98-018, 20 Pages, 1998/03
no abstracts in English
Tanaka, Tadao; Ogawa, Hiromichi
JAERI-Conf 99-004, p.654 - 661, 1998/03
no abstracts in English
Imai, Hisashi*; Nishida, Kaoru*; *; Amemiya, Kiyoshi*; Lin, Weiren*
PNC TJ1449 98-004, 231 Pages, 1998/02
None
 D)+ethylene reaction
D)+ethylene reactionTakayanagi, Toshiyuki; Kurosaki, Yuzuru*; Sato, K.*; *
Journal of Physical Chemistry A, 102(50), p.10391 - 10398, 1998/00
Times Cited Count:29 Percentile:66.76(Chemistry, Physical)no abstracts in English
*; Nagasaki, Shinya*; *; Tanaka, Satoru*; Suzuki, Atsuyuki*; Tanaka, Tadao; Muraoka, Susumu
Radiochimica Acta, 82, p.239 - 242, 1998/00
no abstracts in English